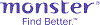Average salary: SG$6,500 /monthly
More statsGet new jobs by email
Search Results: 93 vacancies
- ...Responsibilities: Develop and manage engineering team to meet the demands of design engineering and continued product quality, performance delivery and technology Direct, plan and co-ordinate complex project engineering assignments entailing technical specifications...
- ...Position Summary Joining Nestlé means you are joining the largest Food and Beverage Company in the world. At our very core, we are a... ...delivery of materials to ensure timely delivery for production. To manage day-to-day materials requirement planning (MRP) for factory and...
- ...Summary Joining Nestlé means you are joining the largest Food and Beverage Company in the world. At our very core, we are a human... ...for administration, control, documentation, maintenance and management of recipe systems in accordance with MoC procedures. Support...
- ...cost-efficient repair and maintenance processes in compliance with regulations, and define and execute asset strategy and lifecycle management. Vendor & Contractor Management: You will develop contractual strategy and framework to manage vendors / contractors and corresponding...
- ...Logistics Engineer RESPONSIBILITIES Lead and drive for continuous improvement in materials flow, manning, space and inventory management, fleet management. Participate and drive cost reduction solutions Write Standard Operation Procedures & Work Instructions...
- ...process cycle time and improve quality and yield of finished goods. Motivate team to achieve Key Performance Indices (KPI) set by management Lead taskforce to overcome production and quality issues. Any other tasks that are assigned Who are You? Bachelor’s...
- ...today's competitive landscape. Who are we looking for? Under the general supervision of the Engineering/Process Improvement Manager, the incumbent will be responsible for organizing and coordinating through internal and external resources to ensure engineering services...
- ...revision and update. General lab support including housekeeping, equipment maintenance, inventory of supplies, lab supplies ordering, management of glass wares, autoclaving etc. Any other duties as assigned by your Supervisor/Manager. Key requirements: Degree in a...
- ...equipment Generate monthly SPC report for customers Generate COA (LIMS) Generate Monthly scrap report Internal Auditor for Management system (QMS/EMS/OHSAS) Ensure good housekeeping of the laboratory at all times Any other tasks as and when assigned. Able to...
- ...description describes the general nature and level of work performed in this role. It is not intended to be an exhaustive list of all duties, skills, responsibilities, knowledge, etc. These may be subject to change and additional functions may be assigned as needed by management....
- ...Standard Operating Procedures (SOPs), logbooks & Batch Records (BRs) to perform operation Submit samples via Laboratory Information Management System (LIMS) Work duration: 8 months Work hour: 12 hours rotating shift in a 2-2 3-2 2-3 pattern. Day/night shift rotation is...
- ...BMS), Operate and maintain OT systems, tools, and applications. Support IT/OT services including asset inventory, configuration management. Plan, execute, and document calibration and maintenance activities. Conduct risk assessments, obtain permits, and manage...
- ...Job Description Manage shipbuilding projects from construction to completion, ensuring adherence to specifications and timelines. Analyze workflows and implement best practices to streamline construction works, reduce costs, and improve productivity. Ensure compliance...
- ...complying to contract and engineering designs. Reviews construction progress against Project Schedule and report to the Hull delivery manager Ensures that all construction and installations to FPSO are performed according to project Quality control, NDE examiners,...
- ...operations. Collaborate interdepartmentally to drive assigned tasks to completion Perform other tasks assigned by your Lead / Manager. What we are looking for Proven experience in Engineering or Biopharmaceutical processing. Familiarity FDA & EMA GMP and regulatory...
- ...all times. Document results in accordance with Lonza policies and current Good Manufacturing Practices (cGMP). With support from Manager/Study Lead, perform routine downstream operations with a high level of accuracy. Plan and prioritize lab work with input from the...
- ...Documentation : Record maintenance, calibration, and mapping results accurately. Compliance : Adhere to GMP, EHS, and safety standards; manage work permits for external vendors. Issue Reporting : Escalate non-compliance issues, delays, or equipment breakdowns. General...
- ...innovative technology platforms and extensive experience – we, in Singapore are proud to be part of this global network. What you’ll do: Manage incoming and outgoing flows from / to 3PL Locations. Pro-active monitoring shipments to ensure quality and timely deliveries,...
- ...Science Park, works closely with our customers, and with different functions including MSAT, Manufacturing, Regulatory, Proposal/Program Management, Process Development, Supply Chain, Quality Assurance and external testing facility. As Senior Scientist you will involve in...
3700 SGD
...from developing an appreciation of the industry practices, and applicable technology developments. Scope of work may include supplier management, in-process quality control design, outgoing quality controls, quality assurance planning, quality management system maintenance and...