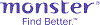Average salary: SG$2,200 /monthly
More statsGet new jobs by email
Search Results: 55 vacancies
- ...in Engineering/Science field including Chemical Engineering, Pharmaceutical Engineering or any other relevant courses Other requirements... ...investment of S$510 million, our state-of-the-art vaccines manufacturing facility at Tuas was opened in June 2009 by Singapore's Prime...
6 SGD per hour
...Trainee Job Title: PCP Trainee- Biotechnologist, Mammalian Manufacturing Department: Mammalian Manufacturing Today, Lonza is a... ...Singapore? We offer you exposure to upcoming biotech and established pharmaceutical companies and to the manufacturing technology needed to...- ...As Equipment Specialist, you will be responsible to produce pharmaceutical products by executing assigned activities according to production... .... Key Responsibilities: Execution of all assigned manufacturing activities according to production schedule and in compliance...
- ...Have good quality mindset and integrity to ensure products are manufactured with the highest quality Participate in deviation... ...engineering Min 2 years of experience in Biologics, Chemical or Pharmaceutical industry Experience in working with GMP Familiar with...
- ...of work we want to be part of. Did you know that we are the 1st biologics contract manufacturer in Singapore? We offer you exposure to upcoming biotech and established pharmaceutical companies and to the manufacturing technology needed to produce mammalian treatments and...
- ...of work we want to be part of. Did you know that we are the 1st biologics contract manufacturer in Singapore. We offer you exposure to upcoming biotech and established pharmaceutical companies and to the manufacturing technology needed to produce mammalian treatments and...
- ...kind of work we want to be part of. Did you know that we are the 1st biologics contract manufacturer in Singapore, We offer you exposure to upcoming biotech and established pharmaceutical companies and to the manufacturing technology needed to produce mammalian treatments...
- ...all activities are carried out in compliance with current Good Manufacturing Practices (cGMP) and safety standards. Working closely... ...experience in a manufacturing environment, preferably in the pharmaceutical or healthcare industry. Strong technical and mechanical aptitude...
- ...assigned by Supervision. WHAT YOU'LL BRING TO ALCON: Higher Nitec, Nitec, or Diploma in Engineering, Manufacturing, or a related discipline within pharmaceutical manufacturing. Minimum 3-5 years of hands-on experience in pharmaceutical processing, particularly in equipment...
- ...Requirement: At least NITEC in Chemical Process Technology, Chemical Engineering, Pharmaceutical Sciences, Chemical and Life Sciences or equivalent At least 2 years of experience in manufacturing environment, production operations, GMP/GDP manufacturing or any other related...
- ...conducting audits and supporting regulatory inspections within a manufacturing environment. This role requires close collaboration with... ...a related discipline. Minimum 5 years of experience in a pharmaceutical or medical device manufacturing environment, particularly in...
- ...Assurance/Microbial contamination control across the entire Lonza manufacturing network (cross-divisional: Biologics, Cell & Gene Therapy,... ...aseptic / sterile manufacturing and Quality Assurance in the pharmaceutical industry Proven hands-on experience in sterility assurance...
- ...Life Science discipline with a focus in Biologics/ Chemical/ Pharmaceutical/ Engineering Other requirements: Must be enrolled at a SG... ...an investment of S$510 million, our state-of-the-art vaccines manufacturing facility at Tuas was opened in June 2009 by Singapore's Prime...
- ...& Maintenance – Maintenance, troubleshooting, and repair of manufacturing equipment Detailed job descriptions for each area are included... ...experience in a manufacturing environment, preferably in the pharmaceutical or healthcare industry Strong technical and mechanical...
- ...records for January each year. What we’re looking for: Diploma and/or NITEC holders with experience working in pharmaceutical industry / manufacturing fields. Strong knowledge and experience in SAP. Knowledge in Supply Chain management / Logistics will be advantageous...
- ...and propose solutions Essential Requirements: Preferred: Previous experience working in a laboratory environment in the pharmaceutical industry (quality assurance, production), aseptic technique. Administrative activities and GMP and HSE-compliant, efficient...
- ...WHAT YOU'LL BRING TO ALCON: Bachelor in Science/ Engineering field Min 1-2 years of working experience in a medical device or pharmaceutical industry in a Quality function, preferably in training HOW YOU CAN THRIVE AT ALCON: Career Growth & Development – Gain...
- ...winning products with reliable long-term performance. The cornerstone for REC's strong reliability is advanced and highly efficient manufacturing using Industry 4.0 practices. Founded in 1996 in Norway, REC has always been committed to a low carbon footprint in its solar...
- ...electronics, mobile devices, telecom, data storage, and infrastructure, MacDermid Alpha Electronics Solutions has earned the trust of manufacturers worldwide. Our comprehensive range of high-quality solutions and technical services covers the entire electronics supply chain,...
- ...to the global energy industry. Job Duties Job Description This position is responsible for creating accurate and routine manufacturing sequences whereby raw materials and assemblies are processed into quality finished products using the most economical and efficient...